

Focus on testing, inspection, certification, research and development, and strive to be a world-class testing, inspection, certification, research and development institute

The competitive edge from focused and differentiated services, to provide high-quality and full scale technical services
查看全部

The competitive edge from focused and differentiated services, to provide high-quality and full scale technical services


The competitive edge from focused and differentiated services, to provide high-quality and full scale technical services


The competitive edge from focused and differentiated services, to provide high-quality and full scale technical services
查看全部
 Validation of Cleaning, Disinfection & Sterilization Process for Medical Devices
Validation of Cleaning, Disinfection & Sterilization Process for Medical Devices 

View panoramic VR lab

One-stop service provider
Improve efficiency by 30%
Zero-distance special service

Cleaning→disinfection→drying→sterilization

Full Dimensions, Entire Process, Life cycle & One-stop
Metrology & calibration
Certification & audit
R & D analysis
Testing & inspection
Quality management
Reasonable suggestions
Digital, Intelligent Testing & Inspection
Intelligent laboratory
Digital quality
Internet e-commerce platform
Electronic certificate
Integration services, etc.
Common Development and Sharing of Data, Technology & Resource
New material, new energy
Biological engineering
Environmental protection & energy saving
Marine ecology
High-end equipment manufacturing
Artificial intelligence
Internet, Capital Crossover Integration & Innovation
Service network layout
Diversified business
Merger & acquisition
Cooperative partner
Customized service
Solution-based services

Standard Group has built a comprehensive service system with primary focuses on biomedical and life sciences. We are solid developing in ecological environment, comprehensive trade, innovative services and other micron sectors. We have CMA, CNAS qualifications, import and export commodity inspection and appraisal, engineering design, building construction, water conservancy project quality inspection and other qualifications.
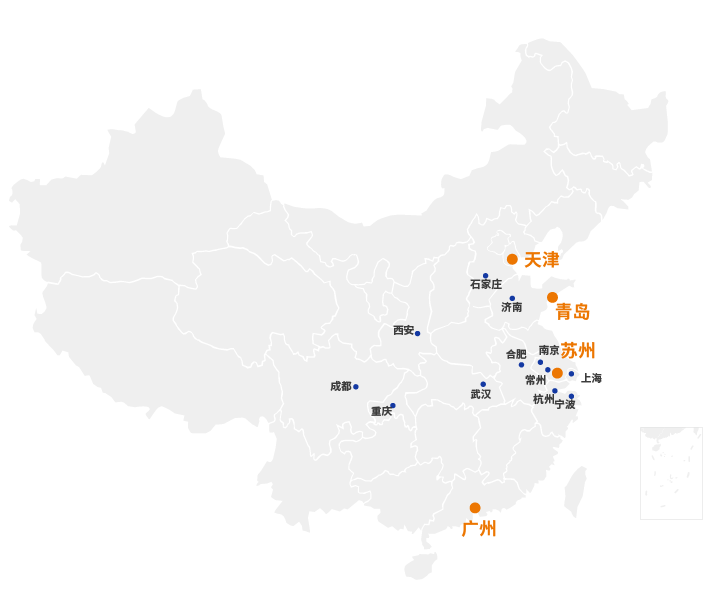

Standard Group laboratories have deployed cross all geographic of China, with 16 branch offices to respond our customers’ inquiries quickly at any time. We determinate to provide close-range and localized professional services at our customers’ conveniences.